October 7, 2019 | Jola Glotzer
Of cocaine and estrogen
Or, why women are more susceptible than men to develop cocaine addiction — two CBC community members Neil Kelleher and Paul Thomas, NU, help to elucidate in a recent Journal of Proteome Research publication
One of the puzzling facts about cocaine addiction is that it is more prevalent among women than among men and it appears to be due to physiological reasons linked to gender. The female hormone — estrogen — has been implicated as the culprit responsible for the observed enhanced rewarding action of the drug in women. However, the exact manner by which estrogen potentiates cocaine effects at the cellular and molecular levels is still unknown.
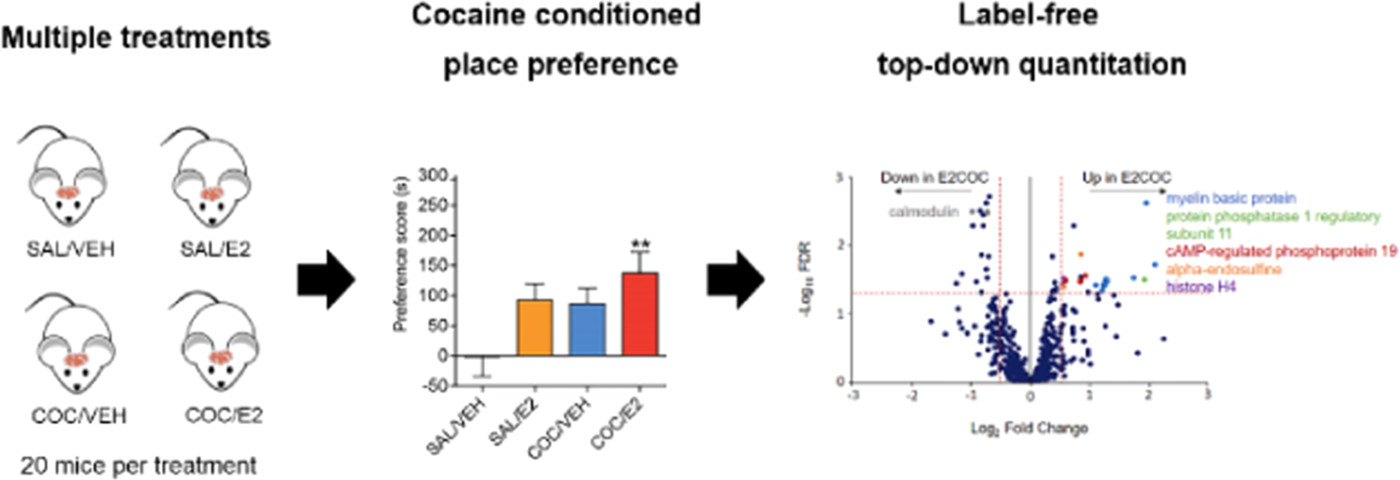
In a recent Journal of Proteome Research publication, a team of Illinois scientists from NU, UIC and UICU explored the molecular changes in female brains of the cocaine addiction mouse model system. Using quantitative top-down proteomics, the researchers identified multiple myelin proteoform modifications in certain regions of the brain, suggesting possible cocaine-induced changes within the white matter. Additionally, several other proteins were identified as affected by the drug. Implications for further directions in studies of estrogen role in the brain signaling pathways as well as in developing intervention treatments for cocaine-addicted females are also discussed.
Two CBC affiliates contributed to the paper: Neil Kelleher and Paul Thomas, NU. Kelleher is the Walter and Mary Elizabeth Glass Professor of Chemistry, Molecular Biosciences, and Medicine at NU. He is also recognized as a CBC Senior Investigator due to his recruitment to NU in 2010 with help from the CBC Recruitment Resources Funds Award. Kelleher is also director of Northwestern Proteomics, an NU-based facility that has evolved from the Proteomics Center of Excellence (PCE), which was originally established with the CBC support. Kelleher’s additional CBC awards and community contributions are listed below the article. The CBC is proud to have played a role in Kelleher’s recruitment and to have helped support his research over the years.
Thomas is Associate Director of Northwestern Proteomics and Research Associate Professor, Molecular Biosciences at NU. For six consecutive years (2010-2015), Thomas taught at the CBC Summer Workshops in Proteomics and Informatics.
The CBC congratulates all of the authors involved in the current study.
Publication linked to the *CBC funding:
Park HM, Satta R, Davis RG, Goo YA, LeDuc RD, Fellers RT, Greer JB, Romanova EV, Rubakhin SS, Tai R, Thomas PM, Sweedler JV, Kelleher NL, Patrie SM, Lasek AW. Multidimensional Top-Down Proteomics of Brain-Region-Specific Mouse Brain Proteoforms Responsive to Cocaine and Estradiol. J Proteome Res. 2019 Oct 2. [Epub ahead of print] (PubMed)
ABSTRACT:
Cocaine addiction afflicts nearly 1 million adults in the United States, and to date, there are no known treatments approved for this psychiatric condition. Women are particularly vulnerable to developing a cocaine use disorder and suffer from more serious cardiac consequences than men when using cocaine. Estrogen is one biological factor contributing to the increased risk for females to develop problematic cocaine use. Animal studies have demonstrated that estrogen (17β-estradiol or E2) enhances the rewarding properties of cocaine. Although E2 affects the dopamine system, the molecular and cellular mechanisms of E2-enhanced cocaine reward have not been characterized. In this study, quantitative top-down proteomics was used to measure intact proteins in specific regions of the female mouse brain after mice were trained for cocaine-conditioned place preference, a behavioral test of cocaine reward. Several proteoform changes occurred in the ventral tegmental area after combined cocaine and E2 treatments, with the most numerous proteoform alterations on myelin basic protein, indicating possible changes in white matter structure. There were also changes in histone H4, protein phosphatase inhibitors, cholecystokinin, and calmodulin proteoforms. These observations provide insight into estrogen signaling in the brain and may guide new approaches to treating women with cocaine use disorder.
Featured CBC Community member(s):
Neil Kelleher, NU
- CBC Exploratory Workshop (2013):
▸ The CBC Exploratory Workshop on Cellular Heterogeneity
Neil Kelleher (NU) – Workshop Organizer and Speaker - CBC Science Day (2011):
Neil Kelleher (NU) – Science Day Speaker - CBC Catalyst Award (2010):
▸ Phosphoproteomic Analysis of NADPH Oxidase Activation
PIs: Neil Kelleher (NU) and Richard Ye (UIC) - *CBC Recruitment Resources Award (2010):
▸ CBC Senior Investigator
PI: Neil Kelleher (NU) - CBC Seminar (2010):
▸ Top Down Mass Spectrometry: Has Its Decade Now Come?
Neil Kelleher (NU, UIUC then) – Seminar Speaker
Paul Thomas, NU
ARTICLES PUBLISHED IN THE PAST ABOUT THE FEATURED CBC COMMUNITY MEMBER(S):
October 1, 2019
▸ Ordering proteoforms
In a recent issue of Nature Methods, CBC Senior Investigator Neil Kelleher, NU, proposes a five-level classification system for proteoform identifications
August 6, 2019
▸ New oncogenic driver discovered
A CBC Lever Award plus a quartet of CBC awardees: Jon Oyer, Jonathan Licht and Neil Kelleher, NU, and Alex Ruthenburg, UChicago, contribute to a recent Cancer Discovery publication
July 8, 2019
▸ Defining the NSD2 interactome
Two CBC Awardees, Jonathan Licht and Neil Kelleher, identify posttranslational modification crosstalk that may play a role in carcinogenesis
July 8, 2019
▸ New insight into liver cancer treatment
CBC Awardee Rick Silverman and a CBC Senior Investigator, Neil Kelleher, NU, collaborate on developing therapeutics for hepatocellular carcinoma (HCC)
June 17, 2019
▸ Copper centers revealed with top-down mass spectrometry
Three CBC Awards contribute to today’s publication in Nature Communications!
May 13, 2019
▸ Battling epigenetic lymphomas
CBC Senior Investigator Neil Kelleher, NU, contributes to a new publication identifying a potential novel therapeutic pathway to treat the so called EZH2 dysregulated lymphomas
May 7, 2019
▸ Proteoforms explained
A recent review in Proteomics, co-authored by a CBC Senior Investigator and proteomics expert, Neil Kelleher, NU
August 9, 2018
▸ Top-down Proteomics
CBC Senior Investigator, Neil Kelleher, NU, explains the advantages of “top-down” versus “bottom-up” proteomics in early cancer detection and progression
November 13, 2017
▸ New insights into regulation of gene expression: work from the Shilatifard lab (NU) with contributions from two CBC scientists, Neil Kelleher and Jeffrey Savas.
October 5, 2017
▸ CBC Senior Investigator, Neil Kelleher, NU, deciphers molecular assembly of a gut toxin, colibactin
February 9, 2016
▸ “Clasping” Collaboration
Three CBC Scientists Join Forces to Develop Exceptional-Quality Antibodies Displaying an Unprecedented Mode of Action