September 27, 2019 | Jola Glotzer
Hypothalamus, the primary responder to the light–dark cycle
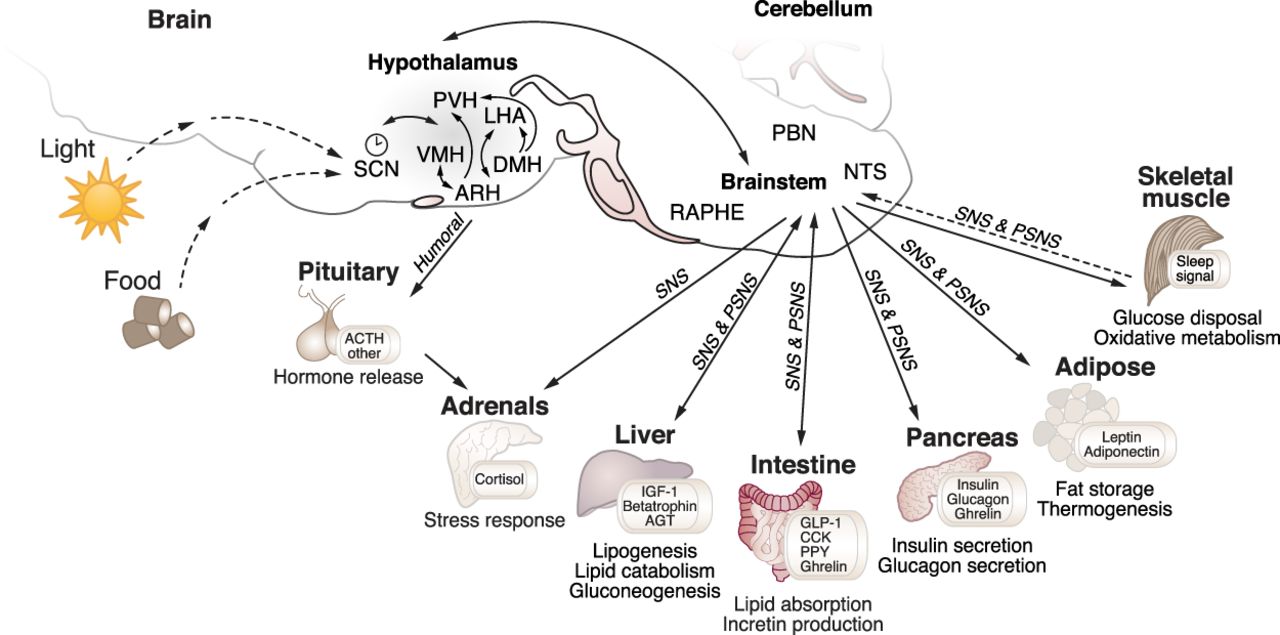
In a recent Genes & Development review, CBC Spark Award recipient Joseph Bass, NU, explains what is known about the interplay between the circadian rhythm and the hypothalamus, and their role in regulating metabolism

Joseph Bass, MD, PhD, the Charles F. Kettering Professor of Medicine in the Division of Endocrinology, director of the Center for Diabetes and Metabolism, NU
A comprehensive and up-to-date review addressing an important function of the hypothalamus as both a primary responder to the light–dark cycle and a master upstream regulator of metabolism was recently published in Genes & Development, “Neurogenetic basis for circadian regulation of metabolism by the hypothalamus.” Senior author on the review is Joseph Bass, MD, PhD, the Charles F. Kettering Professor of Medicine in the Division of Endocrinology and Director of the Center for Diabetes and Metabolism, NU.
The publication acknowledged multiple funding sources including a CBC Spark Award that Bass received in 2011 together with Nissim Hay, UIC, Graeme Bell and Louis Philipson, UChicago. Bass was also a co-recipient of an earlier CBC Catalyst Award (2007).
The CBC applauds Bass for his long-standing efforts in conducting research supported by the CBC Spark Award funds!
Publication attributed to the *CBC funding:
Cedernaes J, Waldeck N, Bass J. Neurogenetic basis for circadian regulation of metabolism by the hypothalamus. Genes Dev. 2019 Sep 1;33(17-18):1136-1158. Review. (PubMed)
ABSTRACT:
Circadian rhythms are driven by a transcription–translation feedback loop that separates anabolic and catabolic processes across the Earth’s 24-h light–dark cycle. Central pacemaker neurons that perceive light entrain a distributed clock network and are closely juxtaposed with hypothalamic neurons involved in regulation of sleep/wake and fast/feeding states. Gaps remain in identifying how pacemaker and extrapacemaker neurons communicate with energy-sensing neurons and the distinct role of circuit interactions versus transcriptionally driven cell-autonomous clocks in the timing of organismal bioenergetics. In this review, we discuss the reciprocal relationship through which the central clock drives appetitive behavior and metabolic homeostasis and the pathways through which nutrient state and sleep/wake behavior affect central clock function.
ACKNOWLEDGMENTS:
We thank Biliana Marcheva for illustrations, Kathryn Moynihan Ramsey for help with the manuscript, and Ravi Alada, Raj Awatramani, Talia Lerner, and Jim Surmeier for helpful discussions. This research was supported by National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) grants R01DK090625, R01DK113011, and R01DK100814; National Institute on Aging grant P01AG011412; Chicago Biomedical Consortium S-007; and University of Chicago Diabetes Research and Training Center grant P60DK020595 (to J.B.). J.C. was supported by the Swedish Society for Medical Research and the Swedish Brain Research Foundation. N.W. was supported by the T32 GM008061 training grant.
Featured CBC Community member(s):
Joseph Bass, NU
- *CBC Spark Award (2011):
▸ Leptin Peptide in Diabetes: From Mechanism to Therapeutics
PIs: Joseph Bass (NU), Graeme Bell (UChicago) and Nissim Hay (UIC) - CBC Catalyst Award (2007):
▸ Chicago Consortium in Diabetes and Obesity Genetics
PIs: Joseph Bass (NU), Graeme Bell (UChicago), Louis Philipson (UChicago) and Nissim Hay (UIC)
ARTICLES PUBLISHED IN THE PAST ABOUT THE FEATURED CBC COMMUNITY MEMBER(S):
September 10, 2019
▸ Circadian rhythm and hunger
Recent work by a CBC Awardee Joseph Bass, NU, featured in the Summer 2019 issue of Northwestern Medicine Magazine, exemplifies the continuity of a research direction initiated by a 2011 CBC Spark Award
August 8, 2019
▸ “Medicine in the Fourth Dimension”
In a recent review in Cell Metabolism, CBC Spark Award recipient, Joseph Bass, NU, discusses the impact of circadian biology on drug development
April 11, 2019
▸ Is it time to eat?
CBC Spark Awardee, Joseph Bass, NU, identifies a subset of neurons in the hypothalamus that coordinate circadian rhythm-dependent sense of hunger
November 8, 2018
▸ NF-kappa beta, inflammation and sleep disorders
CBC Spark Award recipient, Joseph Bass, NU, discovers a genetic link between the causes of inflammation and the circadian clock control
October 31, 2016
▸ Muscles Have Circadian Clocks that Control Exercise Response
The time of day determines a muscle’s energy efficiency and metabolic response
November 16, 2015
▸ Circadian Clock Controls Insulin and Blood Sugar in Pancreas
Map of thousands of genes suggests new therapeutic targets for diabetes